The CIP code (code identifiant de présentation) is a unique numerical identifier assigned to each authorized pharmaceutical presentation in France. It allows a drug to be precisely identified based on its name, dosage, pharmaceutical form, and packaging the contents of that packaging.
Since Decree No. 2021‑1931 of December 30, 2021, the CIP code has legal recognition, making its assignment and use mandatory for all medicines authorised on the French market.
Assigned by the French National Agency for the Safety of Medicines and Health Products (ANSM) following the registration or marketing authorization (MA) procedure, the CIP code is used throughout the medication supply chain. It facilitates the identification, invoicing, inventory management, and health traceability of health products.
The technical specifications were defined by the decree of 30 December 2021 concerning the procedures for allocating and coding medicines.
The CIP code is distinct from the NL code, which is associated with a marketing authorisation application, and from the CIS code, which is associated with a drug.
In the hospital setting, identification can also be based on the UCD code (Unité Commune de Dispensation), which identifies the smallest unit of medicine dispensed in hospitals. The UCD code is assigned by the ANSM and can only exist if it is linked to a CIP code.
How is the 13-digit CIP code structured?
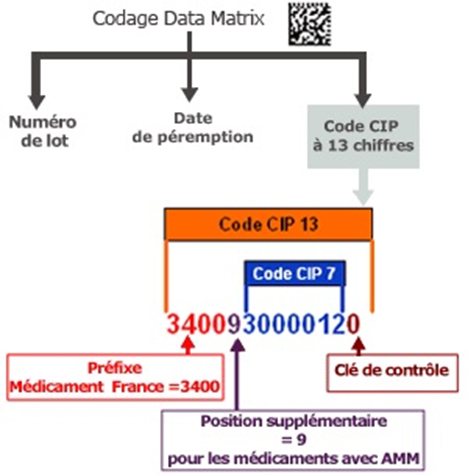
Since January 1, 2009, the 7-digit CIP code has been replaced by a 13-digit code, due to the saturation of the initial format and changes in the regulatory framework. This transition to CIP 13 allows, in particular, the integration of the expiration date and batch number into a machine-readable label, in accordance with stricter health safety requirements.
The 13-digit CIP code is structured as follows:
- “3400”: prefix indicating the pharmaceutical sector in France;
- “9”: heading number designating a pharmaceutical presentation;
- 7 digits: the original 7-digit CIP code, identifying the specific medication presentation (form, dosage, packaging);
- 1 digit: control key, calculated using the EAN 13 algorithm.
When preceded by a “0”, this code complies with ISO/IEC 15459-3 2014 and ISO/IEC 15459-4 2014 standards. This format is compatible with machine-readable barcodes, such as EAN-13 or Data Matrix, placed on packaging and ensuring reliable scanning in pharmacies, healthcare facilities, and specialized software.
In the hospital setting, the UCD code also uses a 13-digit numeric format. The first five digits are “34008,” and the last digit is a check digit calculated using the Luhn algorithm.
What is the CIP code used for?
The CIP code is used to:
- make it easier to manage stocks, supply flows, and market organization.
- help fight against counterfeit medicines.
- identify each medicine clearly, while monitoring marketing authorizations and checking compliance.
- handle reimbursement claims, monitor expenses, and code pricing.
- ensure traceability, monitor safety, and send alerts in case of side effects or product recalls.
Currently, the Inter-Pharmaceutical Club (CIP) is reflecting on the future of drug coding in France. The decision on whether to move to the VIP2400 code will be made by the Ministry of Health.
Article written by Lamya SAOUSSEN, Junior Regulatory Affairs and External Communication Advisor

